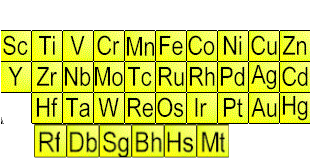
Transition Metals

Name: Scandium
Symbol: Sc
Atomic Number: 21
Atomic Mass: 44.9
Number of Protons/Electrons: 21
Number of Neutrons: 24
Classification: Transition Metals
Discovery: 1879
Discoverer: Lars Nilson
Uses: non
Symbol: Sc
Atomic Number: 21
Atomic Mass: 44.9
Number of Protons/Electrons: 21
Number of Neutrons: 24
Classification: Transition Metals
Discovery: 1879
Discoverer: Lars Nilson
Uses: non

Name: Titanium
Symbol: Ti
Atomic Number: 22
Atomic Mass: 47.8
Number of Protons/Electrons: 22
Number of Neutrons: 26
Classification:Transition Metals
Discovery: 1791
Discoverer: William Gregor
Uses: paint
Symbol: Ti
Atomic Number: 22
Atomic Mass: 47.8
Number of Protons/Electrons: 22
Number of Neutrons: 26
Classification:Transition Metals
Discovery: 1791
Discoverer: William Gregor
Uses: paint

Name: Vanadium
Symbol: V
Atomic Number: 23
Atomic Mass: 50.9
Number of Protons/Electrons: 23
Number of Neutrons: 28
Classification:Transition Metals
Discovery: 1830
Discoverer: Nils Sefstrom
Uses: catalyst, dye, color-fixer
Symbol: V
Atomic Number: 23
Atomic Mass: 50.9
Number of Protons/Electrons: 23
Number of Neutrons: 28
Classification:Transition Metals
Discovery: 1830
Discoverer: Nils Sefstrom
Uses: catalyst, dye, color-fixer

Name: Chromium
Symbol: Cr
Atomic Number: 24
Atomic Mass: 51.9
Number of Protons/Electrons: 24
Number of Neutrons: 28
Classification:Transition Metals
Discovery: 1797
Discoverer: Louis Vauquelin
Uses: Stainless steel
Symbol: Cr
Atomic Number: 24
Atomic Mass: 51.9
Number of Protons/Electrons: 24
Number of Neutrons: 28
Classification:Transition Metals
Discovery: 1797
Discoverer: Louis Vauquelin
Uses: Stainless steel

Name: Manganese
Symbol: Mn
Atomic Number: 25
Atomic Mass: 54.9
Number of Protons/Electrons: 25
Number of Neutrons: 30
Classification:Transition Metals
Discovery: 1774
Discoverer: Johann Gahn
Uses: steel
Symbol: Mn
Atomic Number: 25
Atomic Mass: 54.9
Number of Protons/Electrons: 25
Number of Neutrons: 30
Classification:Transition Metals
Discovery: 1774
Discoverer: Johann Gahn
Uses: steel

Name: Iron
Symbol: Fe
Atomic Number: 26
Atomic Mass: 55.8
Number of Protons/Electrons: 26
Number of Neutrons: 30
Classification:Transition Metals
Discovery: a long time ago
Discoverer: IDK
Uses: steel, hemoglobin
Symbol: Fe
Atomic Number: 26
Atomic Mass: 55.8
Number of Protons/Electrons: 26
Number of Neutrons: 30
Classification:Transition Metals
Discovery: a long time ago
Discoverer: IDK
Uses: steel, hemoglobin

Name: Cobalt
Symbol: Co
Atomic Number: 27
Atomic Mass: 58.9
Number of Protons/Electrons: 27
Number of Neutrons: 32
Classification:Transition Metals
Discovery: 1737
Discoverer: George Brandt
Uses: magnets
Symbol: Co
Atomic Number: 27
Atomic Mass: 58.9
Number of Protons/Electrons: 27
Number of Neutrons: 32
Classification:Transition Metals
Discovery: 1737
Discoverer: George Brandt
Uses: magnets

Name: Nickel
Symbol: Ni
Atomic Number: 28
Atomic Mass: 58.6
Number of Protons/Electrons: 28
Number of Neutrons: 31
Classification:Transition Metals
Discovery: 1751
Discoverer: Alex Cronstedt
Uses: electroplating metal alloys
Symbol: Ni
Atomic Number: 28
Atomic Mass: 58.6
Number of Protons/Electrons: 28
Number of Neutrons: 31
Classification:Transition Metals
Discovery: 1751
Discoverer: Alex Cronstedt
Uses: electroplating metal alloys

Name: Copper
Symbol: Cu
Atomic Number: 29
Atomic Mass: 63.5
Number of Protons/Electrons: 29
Number of Neutrons: 35
Classification:Transition Metals
Discovery: A long time ago
Discoverer: IDK
Uses: electrical conductor
Symbol: Cu
Atomic Number: 29
Atomic Mass: 63.5
Number of Protons/Electrons: 29
Number of Neutrons: 35
Classification:Transition Metals
Discovery: A long time ago
Discoverer: IDK
Uses: electrical conductor

Name: Zinc
Symbol: Zn
Atomic Number: 30
Atomic Mass: 65.3
Number of Protons/Electrons: 30
Number of Neutrons: 35
Classification:Transition Metals
Discovery: 1746
Discoverer: Andreas Marggraf
Uses: metal coating
Symbol: Zn
Atomic Number: 30
Atomic Mass: 65.3
Number of Protons/Electrons: 30
Number of Neutrons: 35
Classification:Transition Metals
Discovery: 1746
Discoverer: Andreas Marggraf
Uses: metal coating

Name: Yttrium
Symbol: Y
Atomic Number: 39
Atomic Mass: 88.9
Number of Protons/Electrons: 39
Number of Neutrons: 50
Classification:Transition Metals
Discovery: 1794
Discoverer: Johann Gadolin
Uses: color TV's
Symbol: Y
Atomic Number: 39
Atomic Mass: 88.9
Number of Protons/Electrons: 39
Number of Neutrons: 50
Classification:Transition Metals
Discovery: 1794
Discoverer: Johann Gadolin
Uses: color TV's

Name: Zirconium
Symbol: Zr
Atomic Number: 40
Atomic Mass: 91.2
Number of Protons/Electrons: 40
Number of Neutrons: 51
Classification: Transition Metals
Discovery: 1789
Discoverer: Martin Klaproth
Uses: nuclear applications
Symbol: Zr
Atomic Number: 40
Atomic Mass: 91.2
Number of Protons/Electrons: 40
Number of Neutrons: 51
Classification: Transition Metals
Discovery: 1789
Discoverer: Martin Klaproth
Uses: nuclear applications

Name: Niobium
Symbol: Nb
Atomic Number: 41
Atomic Mass: 92.9
Number of Protons/Electrons: 41
Number of Neutrons: 52
Classification: Transition Metals
Discovery: 1801
Discoverer: Charles Hatchet
Uses: Non
Symbol: Nb
Atomic Number: 41
Atomic Mass: 92.9
Number of Protons/Electrons: 41
Number of Neutrons: 52
Classification: Transition Metals
Discovery: 1801
Discoverer: Charles Hatchet
Uses: Non

Name: Molybdenum
Symbol: Mo
Atomic Number: 42
Atomic Mass: 95.9
Number of Protons/Electrons: 42
Number of Neutrons: 54
Classification:Transition Metals
Discovery: 1778
Discoverer: Carl Wilhelm Scheele
Uses: aircraft, missiles
Symbol: Mo
Atomic Number: 42
Atomic Mass: 95.9
Number of Protons/Electrons: 42
Number of Neutrons: 54
Classification:Transition Metals
Discovery: 1778
Discoverer: Carl Wilhelm Scheele
Uses: aircraft, missiles
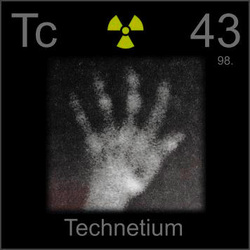
Name: Technetium
Symbol: Tc
Atomic Number: 43
Atomic Mass: 98
Number of Protons/Electrons: 43
Number of Neutrons: 55
Classification:Transition Metals
Discovery: 1937
Discoverer: Carlo Perrier
Uses: radioactive tracing in medicine
Symbol: Tc
Atomic Number: 43
Atomic Mass: 98
Number of Protons/Electrons: 43
Number of Neutrons: 55
Classification:Transition Metals
Discovery: 1937
Discoverer: Carlo Perrier
Uses: radioactive tracing in medicine

Name: Ruthenium
Symbol: Ru
Atomic Number: 44
Atomic Mass: 101.07
Number of Protons/Electrons: 44
Number of Neutrons: 57
Classification:Transition Metals
Discovery: 1844
Discoverer: Karl Klaus
Uses: platinum alloys
Symbol: Ru
Atomic Number: 44
Atomic Mass: 101.07
Number of Protons/Electrons: 44
Number of Neutrons: 57
Classification:Transition Metals
Discovery: 1844
Discoverer: Karl Klaus
Uses: platinum alloys

Name: Rhodium
Symbol: Rh
Atomic Number: 45
Atomic Mass: 102.9
Number of Protons/Electrons: 45
Number of Neutrons: 58
Classification:Transition Metals
Discovery: 1803
Discoverer: William Wollaston
Uses: coatings
Symbol: Rh
Atomic Number: 45
Atomic Mass: 102.9
Number of Protons/Electrons: 45
Number of Neutrons: 58
Classification:Transition Metals
Discovery: 1803
Discoverer: William Wollaston
Uses: coatings

Name: Palladium
Symbol: Pd
Atomic Number: 46
Atomic Mass: 106.4
Number of Protons/Electrons: 46
Number of Neutrons: 60
Classification:Transition Metals
Discovery: 1803
Discoverer: William Wollaston
Uses: jewelry
Symbol: Pd
Atomic Number: 46
Atomic Mass: 106.4
Number of Protons/Electrons: 46
Number of Neutrons: 60
Classification:Transition Metals
Discovery: 1803
Discoverer: William Wollaston
Uses: jewelry

Name: Silver
Symbol: Ag
Atomic Number: 47
Atomic Mass: 107.8
Number of Protons/Electrons: 47
Number of Neutrons: 61
Classification:Transition Metals
Discovery: A long time ago
Discoverer: IDK
Uses: jewelry, photography
Symbol: Ag
Atomic Number: 47
Atomic Mass: 107.8
Number of Protons/Electrons: 47
Number of Neutrons: 61
Classification:Transition Metals
Discovery: A long time ago
Discoverer: IDK
Uses: jewelry, photography

Name: Cadmium
Symbol: Cd
Atomic Number: 48
Atomic Mass: 112.4
Number of Protons/Electrons: 48
Number of Neutrons: 64
Classification:Transition Metals
Discovery: 1817
Discoverer: Fredrich Stromeyer
Uses: poisonous
Symbol: Cd
Atomic Number: 48
Atomic Mass: 112.4
Number of Protons/Electrons: 48
Number of Neutrons: 64
Classification:Transition Metals
Discovery: 1817
Discoverer: Fredrich Stromeyer
Uses: poisonous

Name: Hafnium
Symbol: Hf
Atomic Number: 72
Atomic Mass: 178.4
Number of Protons/Electrons: 72
Number of Neutrons: 106
Classification:Transition Metals
Discovery: 1923
Discoverer: Dirk Coster
Uses: nuclear reactors
Symbol: Hf
Atomic Number: 72
Atomic Mass: 178.4
Number of Protons/Electrons: 72
Number of Neutrons: 106
Classification:Transition Metals
Discovery: 1923
Discoverer: Dirk Coster
Uses: nuclear reactors

Name: Tantalum
Symbol: Ta
Atomic Number: 73
Atomic Mass: 180.9
Number of Protons/Electrons: 73
Number of Neutrons: 108
Classification:Transition Metals
Discovery: 1802
Discoverer: Anders Ekeberg
Uses: capacitors, camera lenses
Symbol: Ta
Atomic Number: 73
Atomic Mass: 180.9
Number of Protons/Electrons: 73
Number of Neutrons: 108
Classification:Transition Metals
Discovery: 1802
Discoverer: Anders Ekeberg
Uses: capacitors, camera lenses

Name: Tungsten
Symbol: W
Atomic Number: 74
Atomic Mass: 183.8
Number of Protons/Electrons: 74
Number of Neutrons: 110
Classification:Transition Metals
Discovery: 1783
Discoverer: Fausto and Juan Jose de Elhuyar
Uses: used widely in electronics industry
Symbol: W
Atomic Number: 74
Atomic Mass: 183.8
Number of Protons/Electrons: 74
Number of Neutrons: 110
Classification:Transition Metals
Discovery: 1783
Discoverer: Fausto and Juan Jose de Elhuyar
Uses: used widely in electronics industry

Name: Rhenium
Symbol: Re
Atomic Number: 75
Atomic Mass: 186.2
Number of Protons/Electrons: 75
Number of Neutrons: 111
Classification:Transition Metals
Discovery: 1925
Discoverer: Walter Noddack
Uses: filaments for mass spectrographs
Symbol: Re
Atomic Number: 75
Atomic Mass: 186.2
Number of Protons/Electrons: 75
Number of Neutrons: 111
Classification:Transition Metals
Discovery: 1925
Discoverer: Walter Noddack
Uses: filaments for mass spectrographs
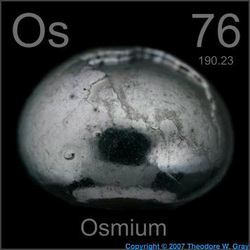
Name: Osmium
Symbol: Os
Atomic Number: 76
Atomic Mass: 190.2
Number of Protons/Electrons: 76
Number of Neutrons: 114
Classification:Transition Metals
Discovery: 1803
Discoverer: Smithson Tenant
Uses: tip gold pen points
Symbol: Os
Atomic Number: 76
Atomic Mass: 190.2
Number of Protons/Electrons: 76
Number of Neutrons: 114
Classification:Transition Metals
Discovery: 1803
Discoverer: Smithson Tenant
Uses: tip gold pen points

Name: Iridium
Symbol: Ir
Atomic Number: 77
Atomic Mass: 192.2
Number of Protons/Electrons: 77
Number of Neutrons: 115
Classification:Transition Metals
Discovery: 1804
Discoverer: S. Tenant
Uses: tip gold pens
Symbol: Ir
Atomic Number: 77
Atomic Mass: 192.2
Number of Protons/Electrons: 77
Number of Neutrons: 115
Classification:Transition Metals
Discovery: 1804
Discoverer: S. Tenant
Uses: tip gold pens
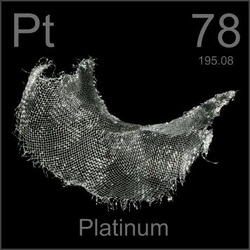
Name: Platinum
Symbol: Pt
Atomic Number: 78
Atomic Mass: 195.07
Number of Protons/Electrons: 78
Number of Neutrons: 117
Classification:Transition Metals
Discovery: 1735
Discoverer: Julius Scaliger
Uses: jewelry, containers
Symbol: Pt
Atomic Number: 78
Atomic Mass: 195.07
Number of Protons/Electrons: 78
Number of Neutrons: 117
Classification:Transition Metals
Discovery: 1735
Discoverer: Julius Scaliger
Uses: jewelry, containers

Name: Gold
Symbol: Au
Atomic Number: 79
Atomic Mass: 196.9
Number of Protons/Electrons: 79
Number of Neutrons: 118
Classification:Transition Metals
Discovery: circa 3000 BC
Discoverer: IDK
Uses: electronics
Symbol: Au
Atomic Number: 79
Atomic Mass: 196.9
Number of Protons/Electrons: 79
Number of Neutrons: 118
Classification:Transition Metals
Discovery: circa 3000 BC
Discoverer: IDK
Uses: electronics
Name: Mercury
Symbol: Hg
Atomic Number: 80
Atomic Mass: 200.5
Number of Protons/Electrons: 80
Number of Neutrons: 121
Classification:Transition Metals
Discovery: A long time ago
Discoverer: LDK
Uses: thermometers
Symbol: Hg
Atomic Number: 80
Atomic Mass: 200.5
Number of Protons/Electrons: 80
Number of Neutrons: 121
Classification:Transition Metals
Discovery: A long time ago
Discoverer: LDK
Uses: thermometers

Name: Rutherfordium
Symbol: Rf
Atomic Number: 104
Atomic Mass: 261
Number of Protons/Electrons: 104
Number of Neutrons: 157
Classification:Transition Metals
Discovery: 1969
Discoverer: Albert Ghiorso
Uses: NON
Symbol: Rf
Atomic Number: 104
Atomic Mass: 261
Number of Protons/Electrons: 104
Number of Neutrons: 157
Classification:Transition Metals
Discovery: 1969
Discoverer: Albert Ghiorso
Uses: NON

Name: Dubnium
Symbol: Db
Atomic Number: 105
Atomic Mass: 262
Number of Protons/Electrons: 105
Number of Neutrons: 157
Classification: Transition Metals
Discovery: 1970
Discoverer: Albert Ghiorso
Uses: Non
Symbol: Db
Atomic Number: 105
Atomic Mass: 262
Number of Protons/Electrons: 105
Number of Neutrons: 157
Classification: Transition Metals
Discovery: 1970
Discoverer: Albert Ghiorso
Uses: Non

Name: Seaborgium
Symbol: Sg
Atomic Number: 106
Atomic Mass: 263
Number of Protons/Electrons: 106
Number of Neutrons: 157
Classification: Transition Metals
Discovery: 1974
Discoverer: Albert Ghiorso
Uses: Non
Symbol: Sg
Atomic Number: 106
Atomic Mass: 263
Number of Protons/Electrons: 106
Number of Neutrons: 157
Classification: Transition Metals
Discovery: 1974
Discoverer: Albert Ghiorso
Uses: Non

Name: Bohrium
Symbol: Bh
Atomic Number: 107
Atomic Mass: 262
Number of Protons/Electrons: 107
Number of Neutrons: 155
Classification: Transition Metals
Discovery: 1976
Discoverer: Peter Armbruster and Gottfried Munzenber
Uses: Non
Symbol: Bh
Atomic Number: 107
Atomic Mass: 262
Number of Protons/Electrons: 107
Number of Neutrons: 155
Classification: Transition Metals
Discovery: 1976
Discoverer: Peter Armbruster and Gottfried Munzenber
Uses: Non

Name: Hassium
Symbol: Hs
Atomic Number: 108
Atomic Mass: 265
Number of Protons/Electrons: 108
Number of Neutrons: 157
Classification:Transition Metals
Discovery: 1984
Discoverer: Peter Armbruster
Uses: Non
Symbol: Hs
Atomic Number: 108
Atomic Mass: 265
Number of Protons/Electrons: 108
Number of Neutrons: 157
Classification:Transition Metals
Discovery: 1984
Discoverer: Peter Armbruster
Uses: Non

Name: Meitnerium
Symbol: Mt
Atomic Number: 109
Atomic Mass: 266
Number of Protons/Electrons: 109
Number of Neutrons: 157
Classification:Transition Metals
Discovery: 1982
Discoverer: Heavy Ion Research Laboratory
Uses: Non
Symbol: Mt
Atomic Number: 109
Atomic Mass: 266
Number of Protons/Electrons: 109
Number of Neutrons: 157
Classification:Transition Metals
Discovery: 1982
Discoverer: Heavy Ion Research Laboratory
Uses: Non